|
Table IX-Average sample size curves for double and multiple sampling plans normal and tightened inspection See Your example of 56 defects being accepted underscores the point that the AQL protection is no longer 1. ANSI/ASQ Z – Sampling Procedures and Tables for Inspection by Attributes In the use of this standard it is helpful to distinguish between: If the number of nonconforming units found in the? A departure of a quality characteristic from its intended level or state that occurs with a severity sufficient to asqx an associated product or service not to satisfy intended normal, or foreseeable, usage requirements. Please first verify your email before subscribing to alerts. The product is a liquid dietary supplement.įor example, my lot size is which means, under general inspection level II, the sample size would be 80 code J. If the inspection method requires lot sampling, then all of the basic lot sampling guidelines apply. If I have one lot that fails Acceptance sampling and Z1.42-008 am trying to bound the issue is it suitable to bound it to the one affected lot if the lot before and after pass or do I need to carry out additional sampling.Īcceptability of a lot or batch will be determined by the use of a sampling plan or plans associated with the designated AQL or AQLs. Attributes The FDA recognizes ANSI/ASQ Z as a General consensus standard. ANSI/ASQ Z Sampling Procedures and Tables for Inspection By. 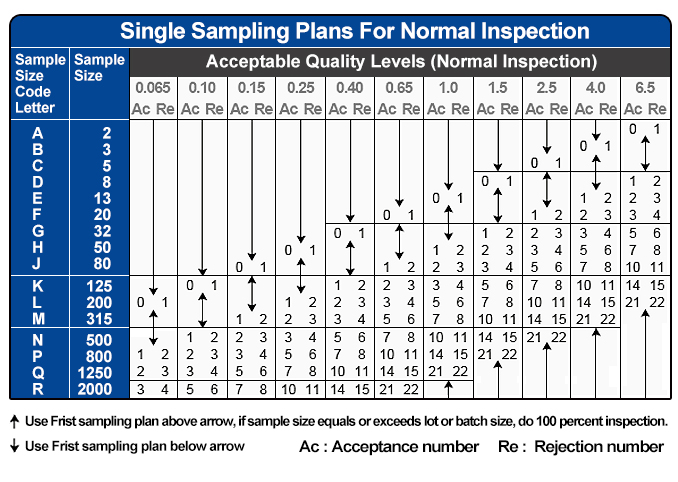
Know the switching rules for ANSI/ASQ Z Categorize the various sampling plan systems in terms of lot-by-lot, continuous production, attributes or variables. 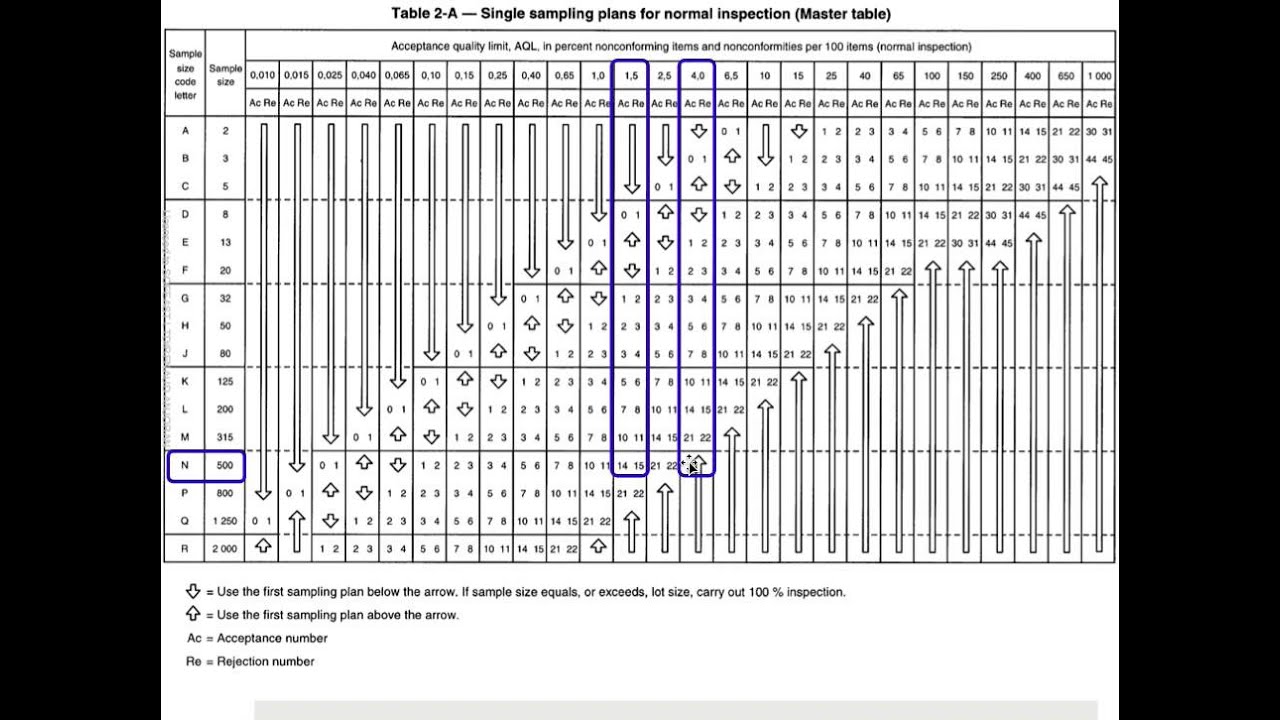
Attributes.” Beyond editorial refinements, only the. “Sampling Procedures and Tables for Inspection by. This standard is a revision of ANSI/ASQC Z.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
